Scientists Advance in Cryopreservation Using Graphene Oxide
Cryopreservation is a procedure where tissues, cells, organs or other biological materials susceptible to damage caused by unregulated chemical kinetics are preserved by cooling to very low temperature, typically ?80 °C or ?196 °C. Relatively low temperature stops any enzymatic or chemical damage to biomaterial, maintaining the genetic and morphological stability. Since the discovery of cryopreservation in 1950s, it has been widely used in the field including medicine, biology and agronomy et al. However, the inevitable ice growth during cryopreserve procedure cause severe ice injuries for the preserved object. The popularization of the commercial cryoprotectant including glycerol and DMSO is depressed due to their low ice recrystallization inhibition (IRI) efficiency and high toxicity. Furthermore, the commercial one causes severe cell dehydration under high concentration.
Recently, due to the fact that antifreeze proteins (AFPs) protect a broad range of biological organisms inhabiting subzero environments, Prof. WANG Jianjun from Institute of Chemistry, Chinese Academy of Sciences (CAS), Prof. FANG Haiping and Dr. WANG Chunlei from Shanghai Institute of Applied Physics investigated the mechanism for how AFPs decrease freezing temperature, inhibiting ice growth and recrystallization. They found that the ordered arrangement of the hydrophobic and hydrophilic functional groups protruding in the ice-binding face facilitated the formation of ice like water, which provided the driving force for binding to the ice surface and inhibited the growth of ice crystal (PNAS, 2016, doi: 10.1073/pnas.1614379114).
Inspired by AFPs, they made use of the unique structure of graphene to investigate the effectiveness of graphene oxide (GO) in controlling ice formation. The results showed that GO inhibited the growth and recrystallization of ice crystal, and shaped ice crystals into a hexagon. Molecular dynamics simulation analysis revealed the formation of stable ice-like water on the surface of GO. Thus, GO formed more hydrogen bonds with ice in comparison with liquid water, which leaded to the formation of a curve and poisoned the continuous growth of the ice lattice due to the Gibbs-Thompson effect.
The application of GO as a cryopreservation agent for horse sperm was also reported for the first time. The results unambiguously showed that GO exhibited a high IRI activity in culture medium of horse sperm and 8.0 mg/mL of NaCl solution. Compared with commercial one, addition of only 0.01% of GO into the culture medium of horse sperm, the motility of horse sperm increased from 24.3% to 71.3%.
This report not only opens a new avenue for the application of 2D materials, but also helps establish a molecular level understanding of AFPs in controlling ice formation. The work has been published on Angew. Chem. Int. Ed.2017, 56, 997-1001.
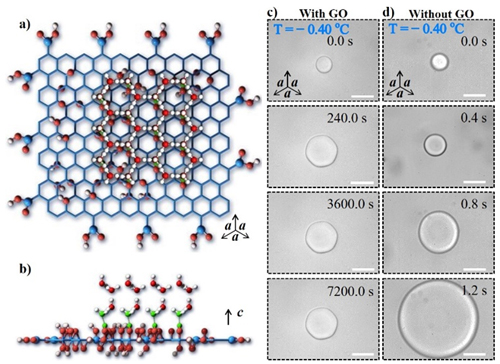
Arrangement of oxidized groups on the basal plane of GO matches well with the ice crystal and effect of GO on the growth and shape of ice crystal. (Image by Prof. WANG Jianjun)
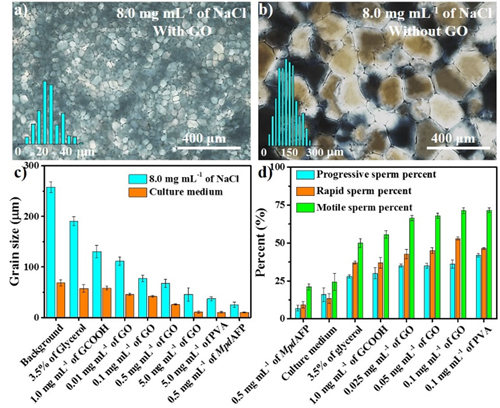
Ice recrystallization inhibition (IRI) activity of GO and its application for cryopreservation (Image by Prof. WANG Jianjun)
Contact:
Prof. WANG Jianjun
Laboratory of Green Printing, Institute of Chemistry, Chinese Academy of Sciences,
Email: wangj220@iccas.ac.cn





