Researchers Realize Direct Conversion of Methane with Carbon Dioxide by Using RhVO3- Cluster Anions
The co-conversion of methane and carbon dioxide to value-added chemicals represents an important subject in C1-chemistry from energy and environmental perspectives. However, the reports dealing with the conversion of CH4 with CO2 were very limited because it is rather difficult to find an active species that could simultaneously activate the reductive CH4 and the oxidative CO2. Design of better-performing catalysts for direct conversion of CH4 with CO2 requires fundamental insights into not only the mechanistic details of the bond-forming and bond-breaking processes, but also the nature of crucial intermediates. Atomic clusters are considered as the intermediate matter to bridge atoms and their bulk counterparts that can act as individual active sites of real-life catalysts.
Recently, a research group from Institute of Chemistry, Chinese Academy of Sciences, has prepared and characterized the first dinuclear cluster species, RhVO3–, which mediates the co-conversion of CH4 and CO2 to oxygenated products, CH3OH and CH2O, in the temperature range of 393-600 K. The resulting cluster ions RhVO3CO– after CH3OH formation can further desorb the [CO] unit to regenerate the RhVO3– cluster, leading to the successful establishment of a catalytic cycle for methanol production from CH4 and CO2 (CH4 + CO2 → CH3OH + CO). The excellent activity of RhVO3– cluster is attributed to its optimal structure: a balance of the concurrent bonding of Rh with the oxygen (O) and metal (V) atoms (O-Rh-V) creates a metallic Rh atom that is highly active for selective methane activation while the reductive Rh?V bond can well reduce the CO2. The molecular-level mechanism of the catalytic CH3OH formation provides insights into the design of active sites that can mediate the co-conversion of methane with soft oxidant CO2.
Their work entitled “Direct Conversion of Methane with Carbon Dioxide Mediated by RhVO3– Cluster Anions” has been published in Angew. Chem. Int. Ed.2019, 58, 17287-17292.
This work was financially supported by the National Natural Science Foundation of China, the Youth Innovation Promotion Association of CAS, and the Beijing Municipal Science & Technology Commission.
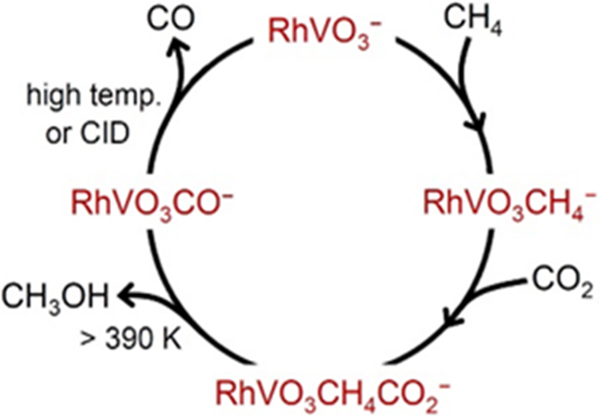
The proposed catalytic cycle for the reaction of CH4 + CO2 → CH3OH + CO mediated by the RhVO3– cluster. (image by YANG Yuan and Dr. ZHAO Yanxia)
Contact:
Prof. HE Shenggui
Institute of Chemistry, Chinese Academy of Sciences
E-mail: shengguihe@iccas.ac.cn





