Researchers Develop Programmable Nanoparticle Platform for Tissue-Specific Protein Degradation
Targeted protein degradation (TPD) has become a transformative approach in both fundamental biology and drug discovery, offering a way to selectively eliminate disease-related proteins. Yet, many current TPD technologies lack precise control over when and where proteins are degraded in the body—capabilities that are critical for safe and effective therapeutic use.
Prof. Ming Wang's group at the Institute of Chemistry, Chinese Academy of Sciences, introduced a programmable platform called Supramolecular Targeting Chimeras (SupTACs) that enables tissue-specific and temporally controlled protein degradation in living organisms.
SupTACs are supramolecular nanoparticles formed through the self-assembly of adamantane-functionalized metal-organic cages and β-cyclodextrin-modified polyethyleneimine. These components interact via host-guest chemistry, allowing SupTACs to co-display target-binding ligands and E3 ligase recruiters on their surface. This multivalent arrangement brings the target protein and the degradation machinery into close proximity, promoting efficient degradation via the ubiquitin-proteasome system.
The researchers demonstrated that SupTACs can selectively degrade proteins in specific tissues, such as the liver and lungs, across multiple species, including non-human primates. For instance, liver- and lung-targeted SupTACs successfully degraded disease-relevant proteins like BRD4 and MAPKs, which are involved in inflammation and cancer.
To highlight the therapeutic potential of the platform, the team designed a SupTAC that targets ACSL4, an enzyme involved in lipid metabolism and ferroptosis. In a mouse model of acute lung injury, lung-specific degradation of ACSL4 significantly reduced ferroptosis and inflammation, suggesting that SupTACs may offer a promising strategy for treating respiratory diseases.
Crucially, SupTACs can also be chemically caged and activated via bioorthogonal reactions, allowing researchers to precisely control the timing of protein degradation. This temporal control adds a new layer of flexibility, enabling researchers to study dynamic biological processes with high precision.
“Our study establishes SupTAC as a programmable and scalable tool for precise protein control in vivo,” said Prof. Wang. “It provides a foundation for exploring complex biological systems and developing new therapeutic strategies.”
By integrating modularity, tissue targeting, and temporal control into a single platform, SupTACs mark a significant advancement in the TPD field-opening new opportunities for both basic research and clinical translation.
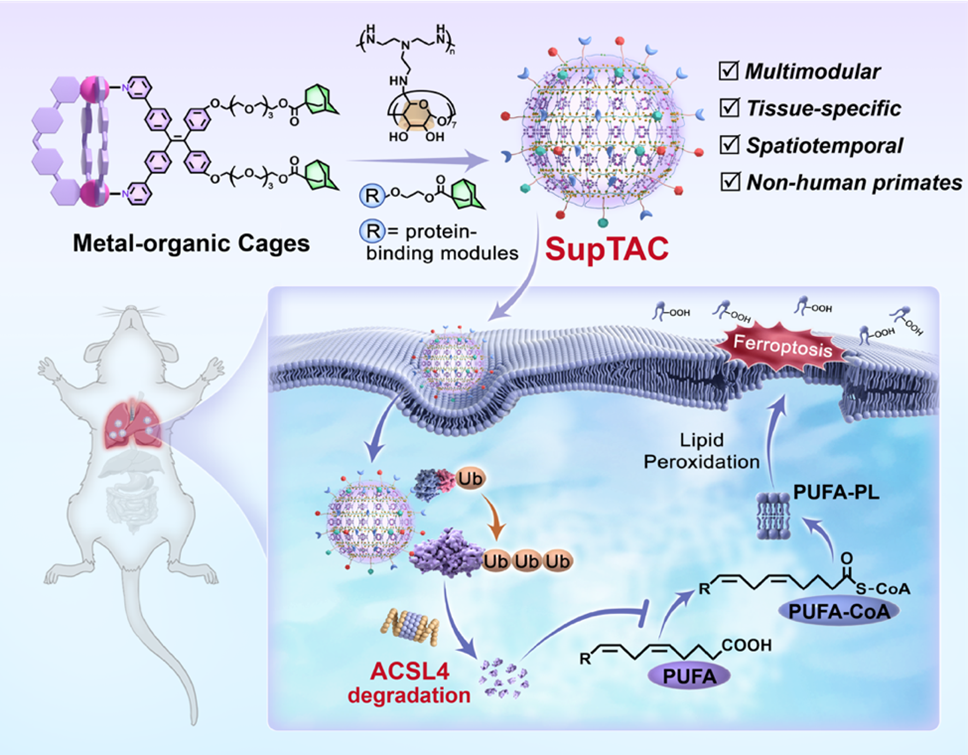
Figure Supramolecular Targeting Chimeras (SupTACs) enables spatiotemporally resolved protein degradation in vivo and ferroptosis suppression acute lung injury
This study was published in cell.
Contact:
Prof. WANG Ming
Institute of Chemistry, Chinese Academy of Sciences
Email: mingwang@iccas.ac.cn





